Sharklet Technologies, Inc. Spearheads the Development of a Standard Test Method for the Evaluation of Contact-Mediated Microbial Transference (E3285-22)
On March 22, 2022, the “Test Method for Evaluation of Contact-Mediated Microbial Transference” led by Sharklet Technologies was officially approved....... Read More
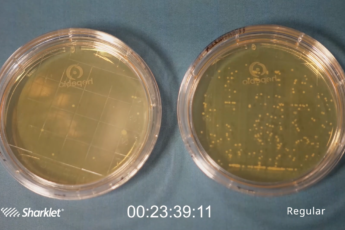
Prepublication: Surface texture limits transfer of S. aureus, T4 Bacteriophage, Influenza B virus and Human coronavirus
Spread of pathogens on contaminated surfaces plays a key role in disease transmission. Surface technologies that control pathogen transfer can...... Read More
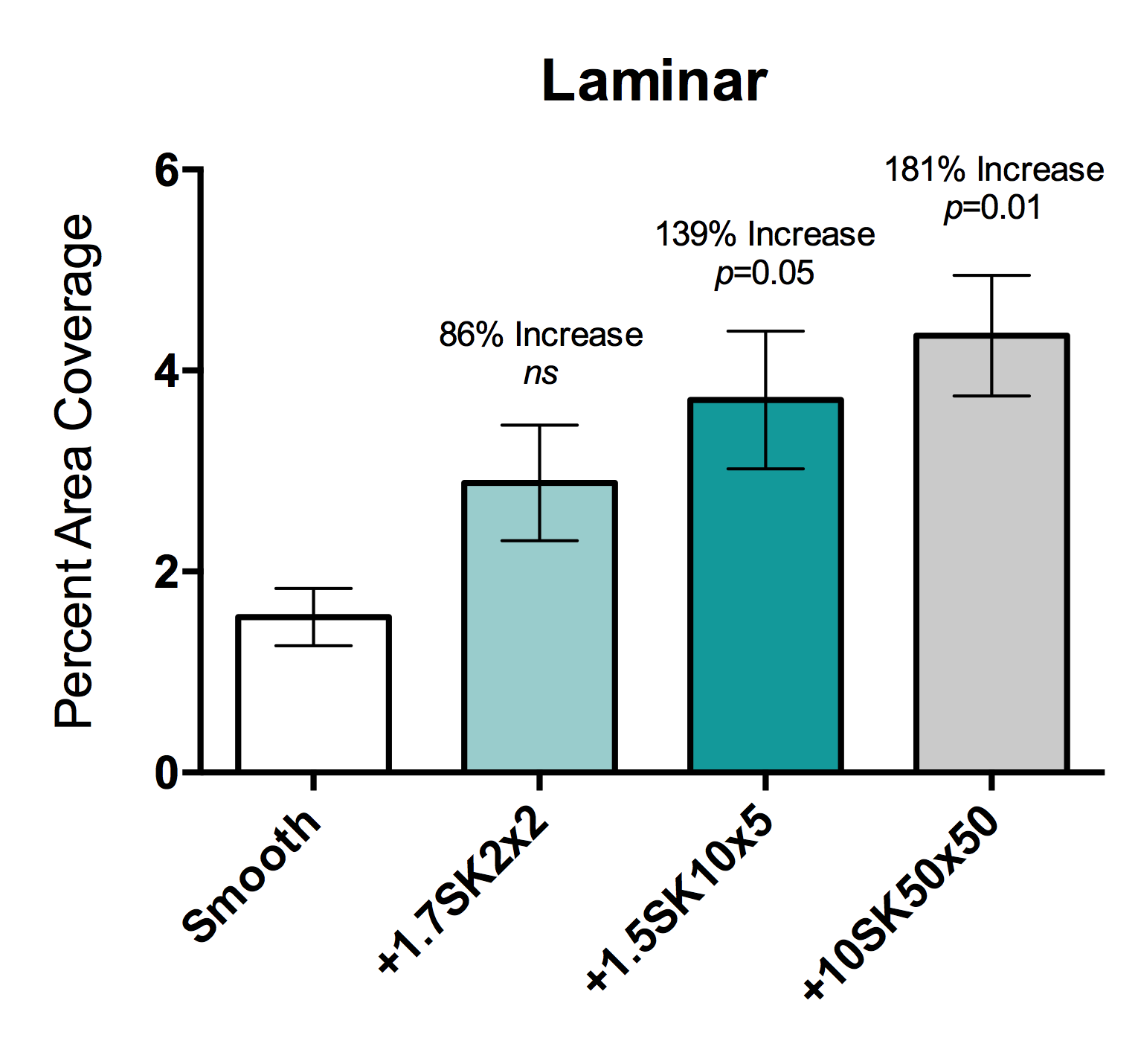
Micropatterns Enhance Endothelial Cell Migration Under Flow Conditions
Cardiovascular disease remains the leading cause of mortality among adults in the US. As a result, nearly 600,000 coronary and...... Read More
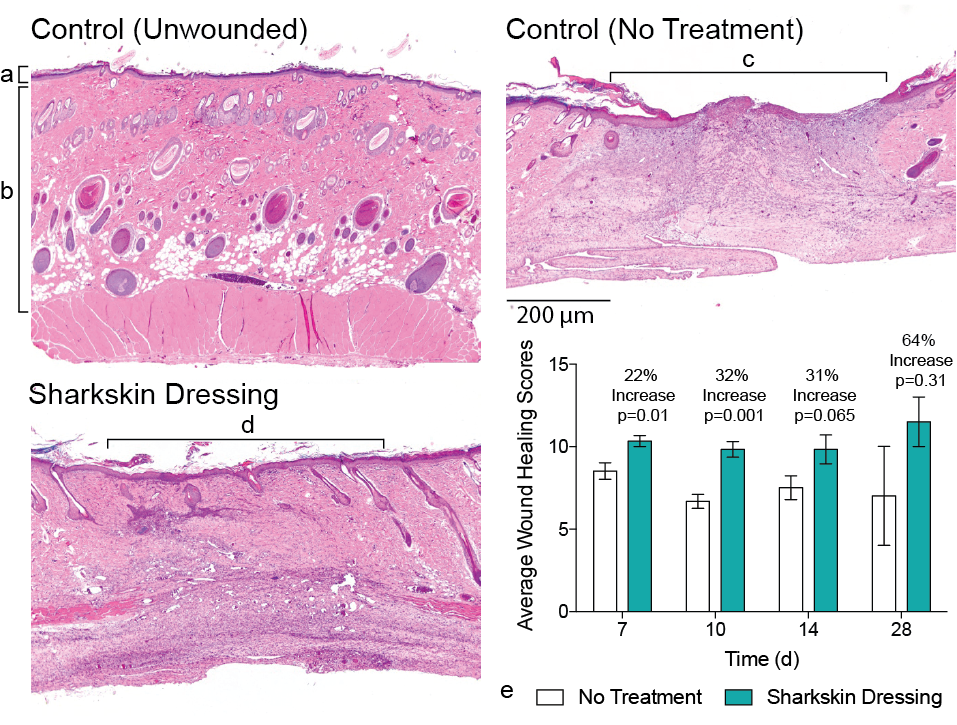
Evaluation of a bilayered, micropatterned hydrogel dressing for full-thickness wound healing
Nearly 12 million wounds are treated in emergency departments throughout the United States every year. The limitations of current treatments...... Read More
Micropatterned Endotracheal Tubes Reduce Secretion-Related Lumen Occlusion
When patients are mechanically ventilated, endotracheal tubes have the potential to disrupt normal airway secretion production and clearance. This ultimately results...... Read More
Micropatterned Protective Membranes Inhibit Lens Epithelial Cell Migration in Posterior Capsule Opacification Model (2015)
During development of the ClearSight™ IOL, Sharklet researched the effect that micropatterned surfaces have on epithelial cells. After surgery to...... Read More
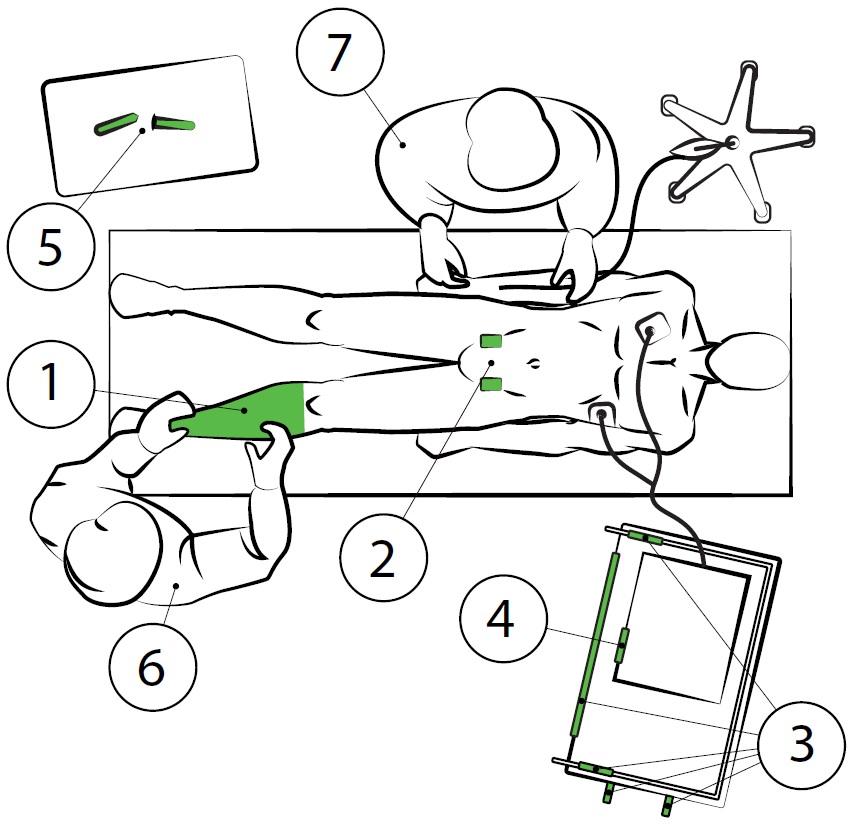
Surface Micropattern Resists Bacterial Contamination Transferred by Healthcare Practitioners (2014)
Sharklet-patterned adhesive films are designed to be deployed in many environments, including hospitals. This study places Sharklet film in key...... Read More
Microtopographies Inhibit Human Lens Epithelial Cell Migration in Posterior Opacification Model (2014)
In this study, Sharklet tested the micropattern’s ability to limit cell migration.... Read More
Bio-Inspired, Engineered Microtopographies Reduce Platelet Adhesion and Activation on Blood-Contacting Materials (2014)
Platelet adhesion and activation are key events in thrombus or clot formation on blood-contacting biomaterials. Thus understanding the complex interactions...... Read More