Sharklet Endotracheal Tube (ETT)
Sharklet uses micropattern alone – no chemicals – to prevent mucus occlusion and control bacterial biofilm on tube surfaces.
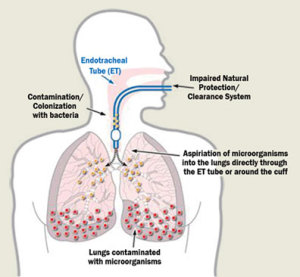
The insertion of an endotracheal tube frequently disrupts the normal secretion and flow of mucus. Mucus gathers in the tube, resulting in a blockage of the airway. This blockage complicates the breathing of the patient, prolonging patient dependence on a ventilator. If the tube occludes completely and cuts off airflow, immediate intervention is required.
Ventilator-associated pneumonia (VAP) is the most common infection in hospital intensive care units. These infections cost hospitals $1.5 billion each year in the United States.
A Sharklet endotracheal tube uses the Sharklet micropattern inside the tube to prevent this dangerous buildup of mucus and biofilm on tube surfaces, and may reduce VAP infections.
The Research
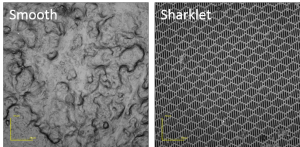
Bacteria adhere to ETTs and form strong, persistent microbial communities. This process allows bacteria to move to the lungs and cause ventilator-associated pneumonia. Sharklet prevents bacterial biofilms as shown in these microscopic images of mucus occlusion on a smooth control surface (left) versus a Sharklet micropatterned surface (right). There is a lack of biofilm buildup on the surface with the Sharklet micropattern.
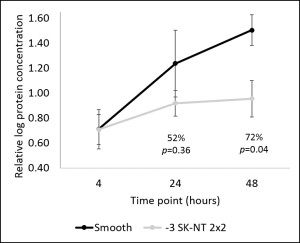
This graph shows protein concentration on smooth and Sharklet surfaces after mucus exposure over 48 hours.
