Pathogen Contamination in Healthcare Settings: Disinfection Is Not Permanent
Environmental cleaning and disinfection are foundational to healthcare infection control. In the United States, guidance and standards are established by the CDC and HICPAC, including the Guideline for Disinfection and Sterilization in Healthcare Facilities and updated best practices for environmental hygiene. These standards require:
- General low-risk areas: Routine disinfection at least once daily
- High-risk and high-touch areas: Disinfection at least twice daily, with additional cleaning as clinically indicated
However, clinical evidence confirms that even strict adherence to disinfection protocols cannot fully eliminate pathogenic bacterial risks in the healthcare environment. Pathogenic bacteria are frequently recovered from high-touch surfaces even after standard disinfection.
A 2024 study published in American Journal of Infection Control, titled Understanding the significance of microbiota recovered from health care surfaces, reported that after routine disinfection, more than 400 samples collected from high-touch surfaces in medical-surgical wards yielded 60 bacterial species, including 18 pathogens[1].
In real clinical practice, surfaces can become recontaminated soon after disinfection, between scheduled disinfection cycles — known as the disinfection gap. During this window, bacteria can recolonize environmental surfaces and spread via contact with healthcare workers’ hands, creating ongoing transmission risks.
Finding effective ways to reduce surface bacterial contamination and transmission without modifying existing disinfection workflows or increasing staff burden has become a critical priority in healthcare infection control.
Mitigation Strategies for the Disinfection Gap
Hand hygiene is recognized as the top priority in infection prevention, as emphasized by the CDC. In clinical care, medical gloves are among the most widely used personal protective equipment, yet they also serve as a major vector for pathogen transmission.
When healthcare workers touch patients, medical devices, and environmental surfaces, gloves can readily pick up and transfer pathogens. Research from the U.S. NIH indicates that 37% of glove-use events involve cross-contamination risks[2].
Recognizing the role of gloves in pathogen transmission, ASTM International’s Committee D11.40 has emphasized that “healthcare-associated infections (HAIs) are a global public health priority, and hands are a primary route for direct and indirect pathogen transmission.”
As early as 2010, the committee specifically established a working group for this purpose to develop an ASTM standard (WK27438) for “antimicrobial medical gloves”, aiming to iterate on ordinary medical gloves, intended to advance conventional gloves to better address clinical needs[3].
Note: The U.S. FDA recognizes and accepts ASTM standards as a basis for regulatory clearance of medical gloves. For example, FDA-cleared nitrile examination gloves must comply with ASTM D6319-19.
Per the ASTM framework, antimicrobial medical gloves should meet three core performance requirements:
- The surface of the glove has antibacterial properties
- The glove reduces microbial exposure to the wearer
- Microbial transfer between surfaces via gloves is limited
Sharklet medical gloves are the only glove product currently capable of meeting all three criteria.
Sharklet nitrile medical gloves contain no added chemical or metallic antimicrobial agents. They rely solely on surface microtopography to inhibit bacterial attachment, colonization, and spread. The technology continuously reduces pathogen transfer between gloves, patients, and environmental surfaces — and provides persistent protection during the disinfection gap to help reduce cross-contamination.
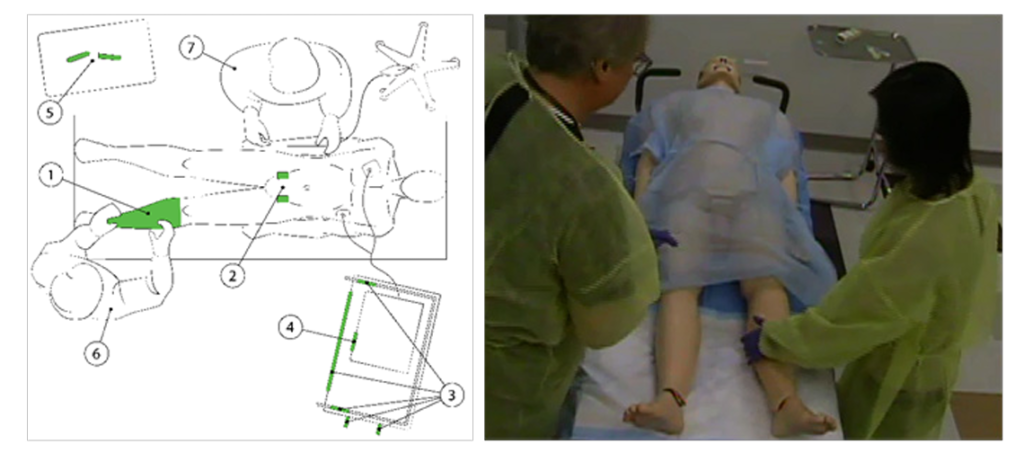
A collaborative study by the University of Colorado Hospital, Center for Biofilm Engineering at Montana State University, and Department of Materials Engineering at the University of Florida was published as Surface micropattern resists bacterial contamination transferred by healthcare practitioners. The study simulated real-world clinical workflows to evaluate Sharklet’s ability to reduce contact transmission.
Results confirmed that, compared to conventional surfaces, Sharklet effectively mitigates transient bacterial contamination in clinical settings and significantly reduces bacterial contact transmission[4], thereby lowering the risk of HAIs.
Globally Recognized Standards Support Sharklet Technology
Beyond strong clinical data and clear market differentiation, Sharklet’s innovative technology is supported by ASTM E3285-22. This established standard framework provides a robust foundation for regulatory acceptance of antimicrobial claims for Sharklet medical gloves worldwide.
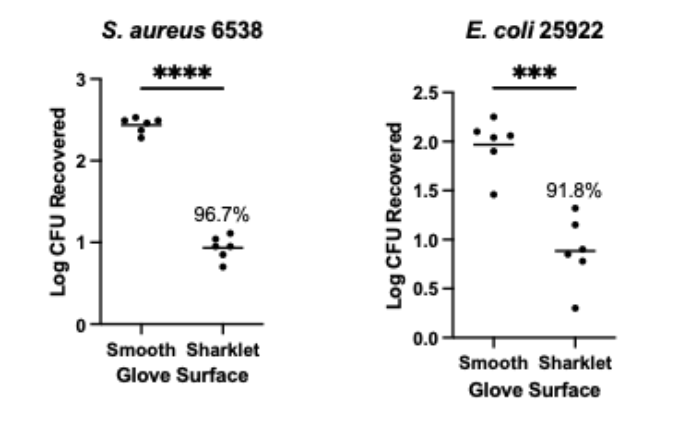
Testing conducted according to ASTM E3285-22 shows that, compared to standard nitrile gloves:
- Sharklet nitrile gloves reduce Staphylococcus aureus contact contamination by 96.7%
- Sharklet nitrile gloves reduce Escherichia coli contact contamination by 91.8%
With full industrial manufacturing development completed, Sharklet medical gloves will soon be commercially available, bringing innovative biomimetic technology to protect the health and safety of healthcare workers and patients worldwide.
